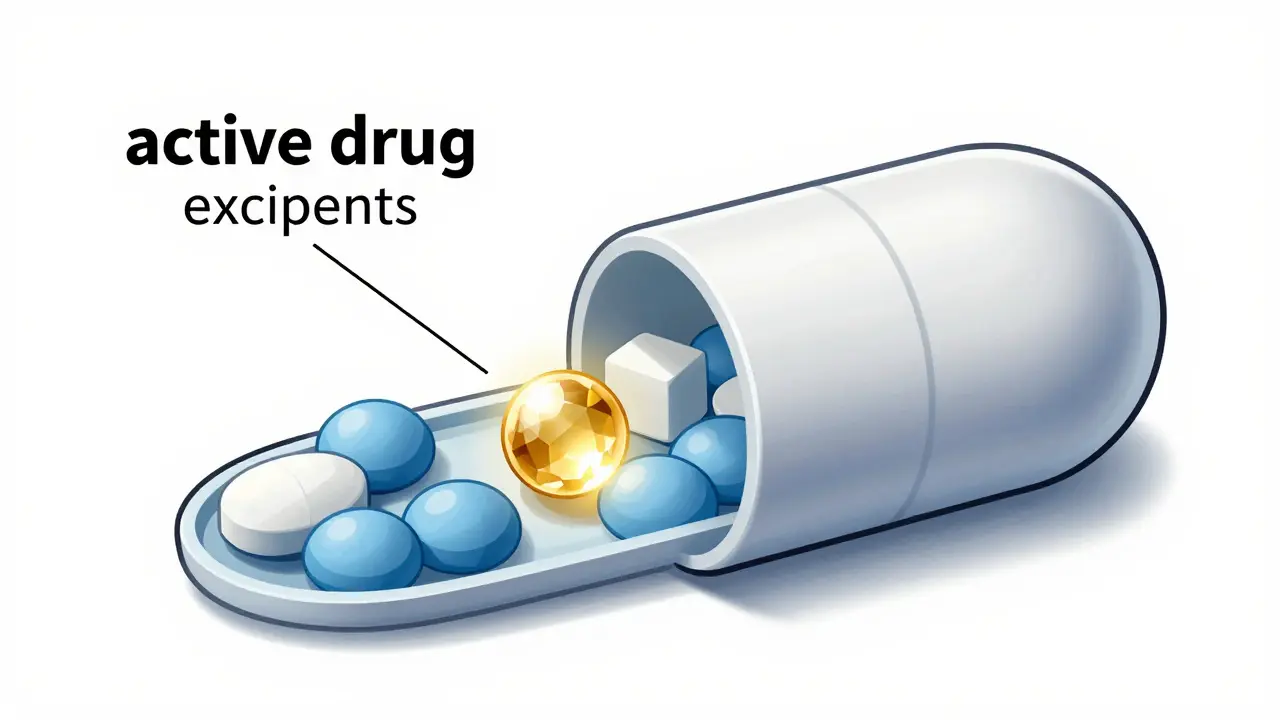
When you look at the back of a medicine bottle, you'll see a list of ingredients. One is the active drug-the part that actually treats your condition. The rest are listed as "inactive ingredients." For years, we've been told these are just fillers, like the flour in a cake, there to hold everything together without changing the result. But is that actually true? Could a different filler in a generic drug change how you feel or how well the medicine works? The short answer is: yes, it can.
While most of these substances are safe, the idea that they are "inactive" is a bit of a misnomer. In reality, Excipients is the technical term for these inactive ingredients, which are components of a drug product other than the active pharmaceutical ingredient (API). They make up anywhere from 60% to 99% of the total mass of a pill. Because they occupy so much of the formula, they aren't just passive bystanders; they actively dictate how a drug dissolves, where it goes in your body, and how long it stays there.
What Excipients Actually Do
If a drug were just the active ingredient, it might be a tiny speck of powder that tastes like battery acid and dissolves instantly in your throat. That's where excipients come in. They serve several practical roles to make the medicine usable for humans:
- Fillers: These add bulk so the pill is large enough to hold. Common examples include Lactose and Microcrystalline Cellulose.
- Binders: These act as the "glue" that keeps the tablet from crumbling. Polyvinylpyrrolidone is a frequent choice here.
- Disintegrants: These ensure the pill actually breaks apart when it hits your stomach. Without them, a pill might pass through you entirely. Croscarmellose Sodium is often used for this.
- Lubricants: These prevent the powder from sticking to the machinery during manufacturing, such as Magnesium Stearate.
- Coatings and Colors: These protect the drug from stomach acid or make it easier to swallow and identify.
Can These "Inactive" Ingredients Actually Be Active?
Here is where it gets interesting. A major study published in Science (2020) flipped the script on the "inactive" label. Researchers found that many substances we've been using for decades actually interact with biological targets in the body. Specifically, out of 314 tested excipients, 38 showed activity against 44 different biological targets. This means they aren't just filling space; they are engaging with your biology.
For example, Propylene Glycol and Sodium Benzoate have shown activity against specific enzymes (monoamine oxidases). While these effects are usually much weaker than the main drug, they can add up. If you're taking multiple medications, each with a handful of "inactive" ingredients, you're essentially taking a cocktail of secondary compounds that may have their own subtle pharmacological effects.
| Excipient | Primary Role | Potential Biological Interaction |
|---|---|---|
| Aspartame | Sweetener | Glucagon receptor activity |
| Propylene Glycol | Solvent/Preservative | Monoamine oxidase A interaction |
| Sodium Benzoate | Preservative | Monoamine oxidase B interaction |
| Lactose | Filler | Digestive distress in intolerant patients |
How This Affects Generic vs. Brand Name Drugs
One of the most common questions is whether a generic drug is "the same" as the brand name. While the Active Pharmaceutical Ingredient (API) must be identical, the excipients don't have to be. This is why a generic pill might be a different color, shape, or taste.
In most cases, the FDA allows different excipients as long as the drug's safety and efficacy remain unchanged. However, for high-risk medications-like those injected directly into the bloodstream (parenteral) or put into the eyes (ophthalmic)-the rules are much stricter. In those cases, the generic must have the exact same inactive ingredients at the exact same concentrations. Why? Because a tiny change in a filler in an eye drop could cause severe irritation or prevent the drug from absorbing properly.
Even for oral pills, changes can matter. For instance, if a manufacturer replaces one disintegrant with another, the drug might dissolve 15% slower. For most people, this is irrelevant. But for a fast-acting medication where every minute counts, that slight shift in dissolution could theoretically impact how quickly you feel relief.
Real-World Risks: When Excipients Cause Problems
Most of the time, you won't notice a difference between excipients. But there are specific scenarios where they become a primary concern:
- Allergies and Intolerances: This is the most common issue. A person with severe lactose intolerance might react poorly to a pill that uses lactose as a filler, even if the actual medicine is safe.
- Unexpected Chemical Reactions: In 2018, several generic versions of the blood pressure drug valsartan were recalled. The issue wasn't the drug itself, but a contaminant called NDMA that formed because of a change in the solvent system used during manufacturing.
- Absorption Changes: Some excipients can change the pH of the environment around the drug, which might make the API absorb faster or slower than intended. This is why some complex generics fail FDA approval; the agency may find that a change in a lubricant like magnesium stearate altered the release rate of the drug too much.
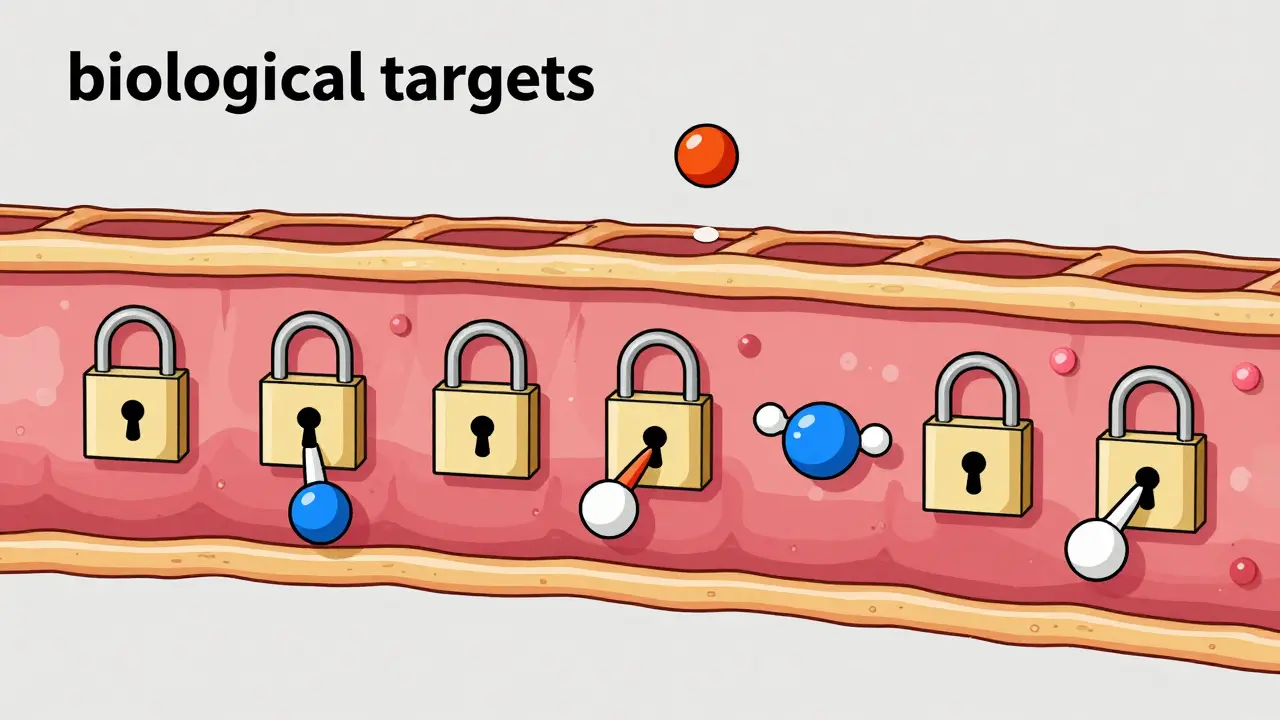
The Future of "Inactive" Ingredients
The pharmaceutical industry is currently in a transition period. We are moving away from the assumption that excipients are inert. The FDA is now looking into computational models to predict how these fillers interact with human targets before the drug even hits the market. There is also a push by the International Pharmaceutical Excipients Council (IPEC) to set clear concentration thresholds. The goal is to define exactly how much of a substance can be used before it stops being "inactive" and starts becoming a pharmacological player.
For the average person, this means that future medications will likely be more tailored. We may see a move toward "clean label" pharmaceuticals where fillers are chosen not just because they are cheap or easy to work with, but because they have the least biological impact on the patient.
Can a change in inactive ingredients make a generic drug less effective?
In rare cases, yes. While the active drug is the same, the excipients control how that drug is released into your system. If a filler or lubricant changes the dissolution rate-meaning the pill dissolves slower or faster than the brand name-it could theoretically affect how quickly or effectively the medicine works, though most generics must prove bioequivalence to be approved.
Why are some inactive ingredients the same in brands but different in generics?
Generic manufacturers often use different fillers or binders because they have different sourcing chains, different manufacturing machinery, or are trying to optimize the cost of production. As long as these changes don't interfere with the drug's performance, regulators like the FDA allow them to be different to encourage competition and lower prices.
Should I be worried about the biological activity of excipients?
For the vast majority of people, no. The concentrations of these substances are very low, and decades of safety data support their use. However, if you have a history of severe allergies or multiple drug sensitivities, it is worth asking your pharmacist for the full list of inactive ingredients to ensure there are no triggers.
What is a "bioequivalence" study?
It is a test where the generic drug and the brand name drug are given to volunteers to see if they reach the same concentration in the blood over the same amount of time. This process ensures that even if the excipients are different, the overall delivery of the active drug is essentially the same.
Which types of medications have the strictest rules for inactive ingredients?
Injected drugs (parenterals), eye drops (ophthalmics), and ear drops (otics) have the strictest requirements. Because these bypass some of the body's natural filters (like the digestive tract), the FDA generally requires the inactive ingredients to be qualitatively and quantitatively identical to the original brand name drug.
Next Steps for Patients and Caregivers
If you're switching from a brand-name drug to a generic and feel a difference in how you respond, don't ignore it. While it's often a placebo effect (the "nocebo" effect, where you expect a generic to work worse), it can occasionally be due to an excipient. Here is how to handle it:
- Check the Label: Compare the "inactive ingredients" list on your old bottle and your new one. Look for common allergens like lactose, gluten, or specific dyes.
- Talk to the Pharmacist: Ask if there is an alternative generic from a different manufacturer. Different companies use different excipients; switching brands of the generic often solves the problem.
- Track Your Symptoms: Note if the reaction is digestive (suggesting a filler issue) or a change in how fast the drug works (suggesting a dissolution issue).