When a brand-name drug hits the market, its manufacturer usually holds a patent that blocks generics for years. But there’s a legal loophole-actually, a carefully designed tool-that lets generic companies challenge those patents before they even make a single pill. This is called a Paragraph IV certification, and it’s one of the most powerful mechanisms in modern drug pricing.
What Is a Paragraph IV Certification?
A Paragraph IV certification is a formal statement filed by a generic drug company when it submits an Abbreviated New Drug Application (ANDA) to the FDA. It says: "One or more patents listed for this drug are invalid, unenforceable, or won’t be infringed by our product." This isn’t just a claim. It’s a legal trigger.
Under the Hatch-Waxman Act of 1984, submitting this certification counts as an "artificial act of infringement." That means the brand-name company can sue the generic firm before the generic drug is sold. Normally, patent holders wait until someone actually copies their product. Here, the system flips that. The generic company has to prove its case upfront.
The goal? Speed up access to cheaper drugs. Without this mechanism, generics might wait years after a patent expires-just because the brand company never sued. Paragraph IV forces the issue.
How the Process Works
The timeline is strict. Once the FDA accepts an ANDA with a Paragraph IV certification, the generic company has 20 days to notify the patent holder. That notice must include the legal and factual reasons why they believe the patent doesn’t apply. No vague statements. No guesswork. Just facts.
Then, the brand company has 45 days to file a lawsuit. If they do, the FDA can’t approve the generic drug for 30 months. That’s the "30-month stay." But here’s the twist: if the court rules in favor of the generic before then, the stay ends early. If the brand company doesn’t sue within 45 days, the FDA can approve the generic right away.
This isn’t a waiting game-it’s a legal race. The clock starts the moment the certification is filed. And the stakes? Billions.
Why It Matters: The 180-Day Exclusivity Prize
The real incentive for generics? The first company to file a successful Paragraph IV certification gets 180 days of market exclusivity. That means no other generic can enter during that time. For a blockbuster drug like Humira or Eliquis, that’s a $500 million windfall.
In 2023 alone, first-filers earned $4.7 billion from this exclusivity period. That’s why companies invest millions in legal teams and patent analysis. It’s not just about making cheaper drugs-it’s about winning a high-stakes financial race.
But here’s the catch: many of these races end in settlements. In 2024, 78% of Paragraph IV cases were settled before trial. Often, the brand company pays the generic to delay launch. These "pay-for-delay" deals cost consumers billions. The FTC filed 17 lawsuits against such agreements in 2023-2024, trying to stop them.

Patent Thicketing: The Brand Company Counterattack
Brand-name companies didn’t sit still. They responded by listing more patents in the FDA’s Orange Book. In 2005, a typical drug had 7.2 patents listed. By 2024, that number jumped to 17.3.
This is called "patent thicketing." It’s not about protecting innovation-it’s about blocking competition. Each patent adds another legal hurdle. A generic company might challenge one patent, only to find three more waiting.
Dr. Aaron Kesselheim from Harvard called it "evergreening." It’s when companies make tiny changes to a drug-like switching the tablet coating or adjusting the dose-and get a new patent. These aren’t breakthroughs. They’re legal tactics.
By 2024, 68% of major branded drugs faced three or more Paragraph IV challenges. That’s not because generics are aggressive. It’s because brand companies made it harder to get past.
Carve-Outs and Skinny Labels: The Smart Workaround
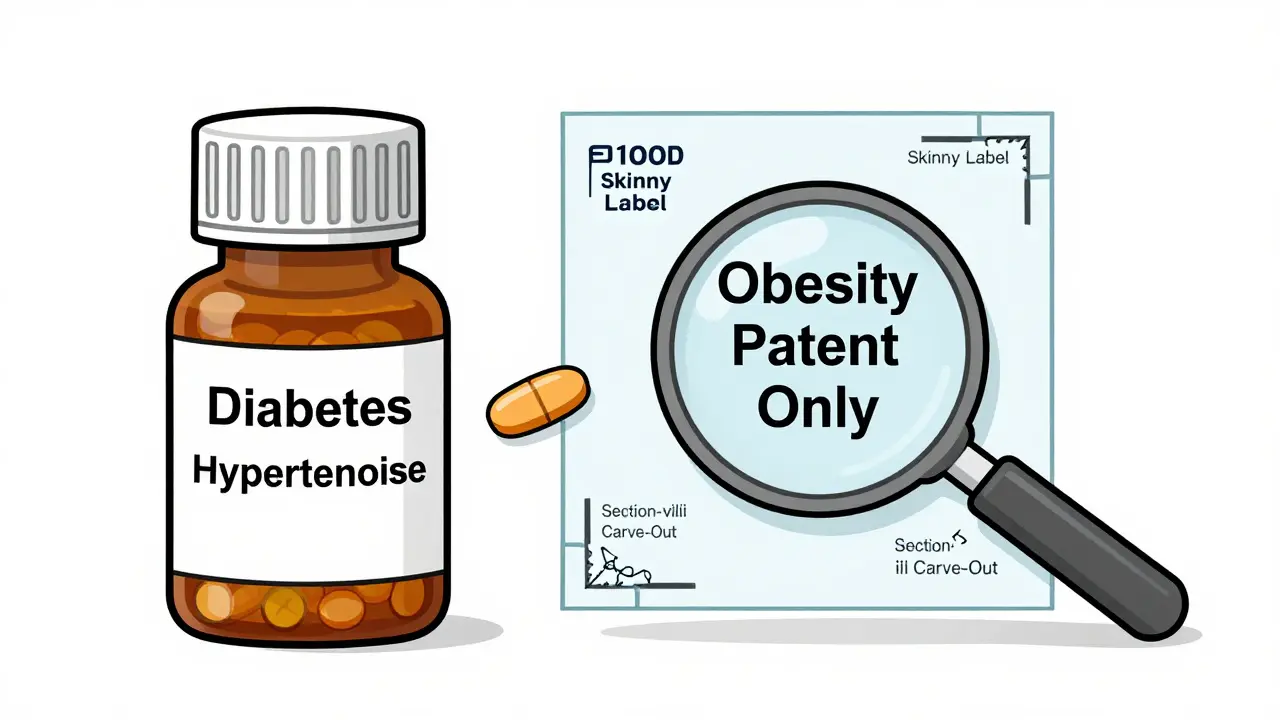
Not every patent covers every use of a drug. Take a medication approved for three conditions: A, B, and C. If only the patent for Condition C is still active, the generic can legally launch a "skinny label"-selling the drug for Conditions A and B only.
This is called a Section viii carve-out. It’s a legal gray area, but it’s allowed. About 37% of Paragraph IV filings in 2023 used this tactic. It lets generics enter the market faster, even if they can’t challenge all patents.
For example, if a drug treats both diabetes and obesity, and the patent only covers the obesity use, the generic can sell it for diabetes alone. No lawsuit. No delay. Just a smarter label.
The Real Cost of Fighting
Going to court isn’t cheap. The average Paragraph IV challenge costs generic companies $12.3 million in legal fees. Cases take nearly 29 months to resolve. That’s why many small firms avoid it entirely.
And if they lose? They lose everything. If the court says the patent is valid, the generic can’t sell the drug until the patent expires. That’s a $100 million+ investment down the drain.
Some try "at-risk" launches-selling the drug before the court decides. In 2024, 22% of generics did this. They risked $217 million in damages if they lost, but they gained $83 million in early sales. For some, the gamble paid off. For others, it was financial ruin.
Who’s Winning? Who’s Losing?
Tea Pharmaceutical led with 147 Paragraph IV filings in 2024. Mylan, Sandoz, and Hikma followed. These are the heavyweights. They have teams of lawyers, pharmacologists, and patent analysts working full-time on this.
On the brand side, AbbVie (Humira), Eli Lilly (Trulicity), and Pfizer (Eliquis) faced the most challenges. These are billion-dollar drugs. The patents are worth fighting over.
But here’s the real story: since 1984, Paragraph IV challenges have saved consumers $2.2 trillion. In 2024 alone, they saved $192 billion. That’s not just corporate profit. That’s money back in patients’ pockets.
What’s Changing in 2025 and Beyond
The FDA updated rules in 2022 to stop generics from "amending" their certifications after a lawsuit starts. That was meant to stop "litigation shopping"-where companies change their claims to delay the process.
And in 2026, the FDA plans to require brand companies to justify every patent they list in the Orange Book. If they can’t prove it’s truly related to the drug, it gets removed. Analysts predict this could cut patent thickets by 30-40%.
Meanwhile, generic success rates are rising. From 2003 to 2019, generics won only 41% of cases. Since 2020, that number jumped to 58%. Why? Supreme Court rulings have narrowed what counts as a patentable invention. That’s helping generics win more often.
The future? More challenges. Fewer patents. Faster access. And more savings.





11 Comments