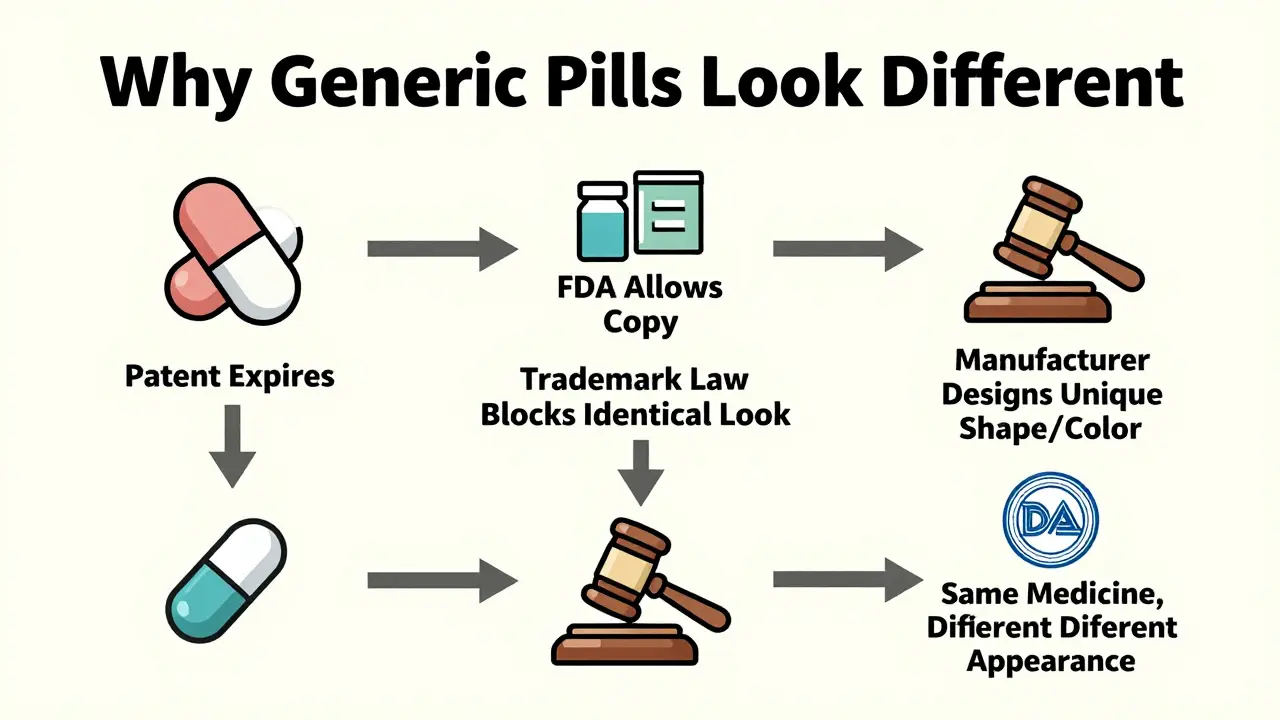
Have you ever picked up your prescription and thought, "This doesn’t look like the last one I got"? You’re not alone. Many people panic when their generic pill changes color, shape, or size. But here’s the truth: that change is legal-and it’s not a mistake. It’s required by trademark law.
Why can’t generic drugs look like the brand name?
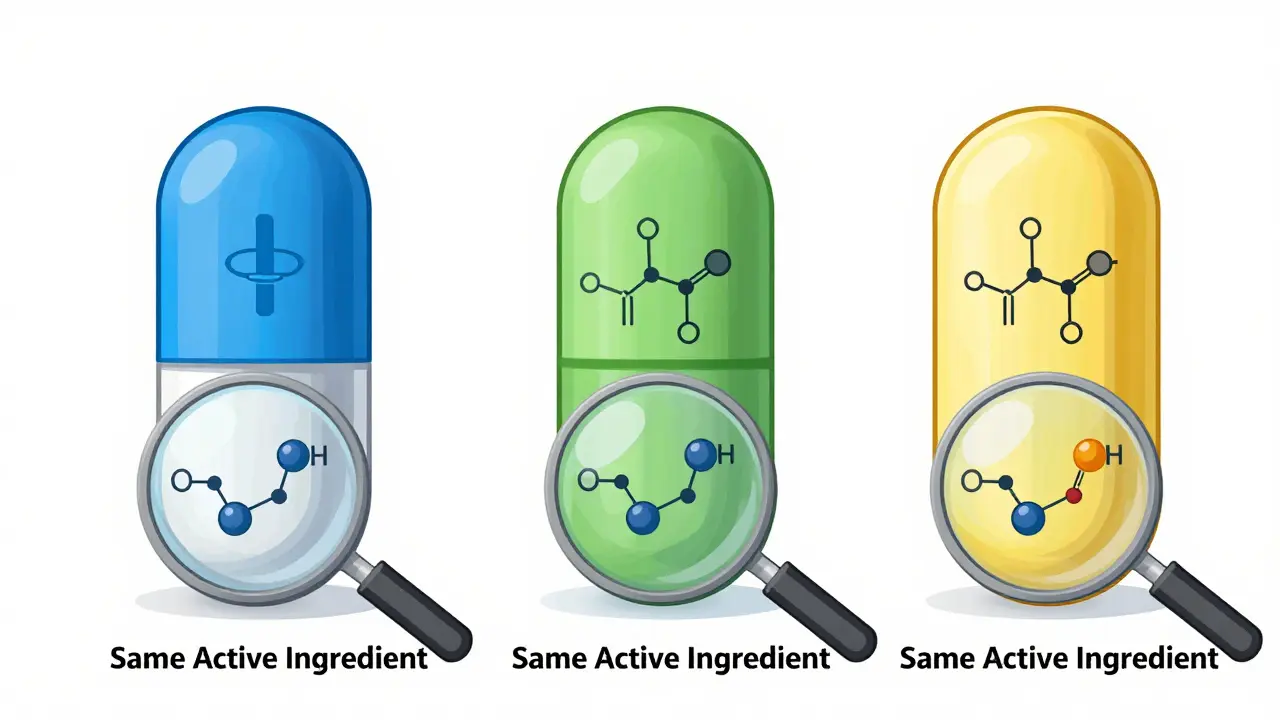
Generic drugs aren’t knockoffs. They’re not fake. They’re not cheaper because they’re weaker. They’re identical in how they work, how safe they are, and how well they treat your condition. The only thing that’s different? How they look.This isn’t random. In the U.S., trademark law says a generic drug cannot look exactly like another drug already on the market. That includes brand-name pills, but also other generics. Why? To prevent confusion. If two pills look identical, a patient might think they’re the same product from the same company. Or worse-they might accidentally take the wrong one.
The FDA makes this clear: "Trademark laws in the United States do not allow a generic drug to look exactly like other drugs already on the market." It’s not about protecting the brand. It’s about protecting you.
What exactly changes-and what doesn’t?
Here’s what stays the same between a brand-name drug and its generic version:
- The active ingredient (the part that treats your condition)
- The strength and dosage
- The way it works in your body
- The safety profile and side effects
- The way it’s taken (tablet, capsule, liquid, etc.)
Here’s what can change:
- Color
- Shape
- Size
- Score lines (the line you break a pill along)
- Flavoring (for liquids or chewables)
- Fillers and binders (inactive ingredients)
These differences don’t affect how the drug works. A 2022 study by U.S. Pharmacist found that generics differ from brand-name drugs by only 3.5% in how much of the drug enters your bloodstream. That’s well within the FDA’s accepted range of 80% to 125%. In other words: your body absorbs it the same way.
How trademark law shapes what you see
Trademark law exists to protect brands. Think Coca-Cola’s red can or Apple’s white earbuds. Those designs are legally protected so no one else can copy them. The same applies to pills. The original drug company spent years designing their pill’s shape and color to make it recognizable. Once the patent expires, other companies can make the drug-but they can’t copy the look.
This forces generic manufacturers to create their own unique appearance. That’s why you might get a blue oval one month and a white capsule the next. Different manufacturers. Different designs. Same medicine.
The FDA actually encourages generic makers to make their pills similar in size and shape to the brand name. Why? To reduce confusion. But they still have to make sure it’s different enough to avoid trademark infringement. It’s a tightrope walk between safety and legality.
What happens when your pill changes again?
Ever refill a prescription and get a totally different-looking pill? That’s not a glitch. It’s normal.
Pharmacies don’t always get the same generic manufacturer each time. One month, your blood pressure pill might come from Teva. The next, from Mylan. Each company uses its own color and shape. That’s why your pill might switch from green to yellow to white-sometimes within the same month.
A 2023 survey by UMass Memorial Health found that nearly half of patients reported confusion or anxiety when their pill changed appearance. Some thought they’d been given the wrong medicine. Others worried it wasn’t as strong.
That’s why pharmacies now put clear labels on vials: "This is a generic version of [Brand Name]." They also train pharmacists to explain the change. Still, mistakes happen. That’s why it’s smart to check the label every time you pick up a refill.
Are generic drugs safe even if they look different?
Yes. Absolutely.
The FDA doesn’t approve a generic drug until it proves it works the same as the brand name. That means testing for:
- How fast the drug enters your bloodstream
- How long it stays in your body
- How well it controls your condition
Manufacturers must submit this data. The FDA reviews it. Then, they inspect the factory. And they keep checking after the drug is on the market.
Dr. Janet Woodcock, former head of the FDA’s drug center, said it plainly: "Generic drug companies must provide scientific evidence that shows their active ingredient is the same as the brand-name medicine." No exceptions.
And the numbers back it up. Over 90% of prescriptions in the U.S. are for generics. Yet they make up only 23% of total drug spending. That’s because they cost 80% to 85% less. Millions of people rely on them every day. And they work.
What should you do if your pill looks different?
Don’t panic. Don’t stop taking it. Do this:
- Check the label. It should say the generic name and the brand name it copies.
- Compare the pill shape and color to the description on the label.
- Ask your pharmacist: "Is this the same medicine I was taking before?"
- If you’re still unsure, call your doctor. But don’t skip your dose.
Pharmacists are trained to handle this. They know appearance changes are normal. They’re also required to alert you if the manufacturer switched. Many pharmacies now include a note on the sticker: "Your medication has changed manufacturers. This is normal."
Why this system works
This whole setup-different looks, same medicine-isn’t broken. It’s designed to do two things:
- Let cheaper drugs into the market after patents expire
- Keep patients from mixing up pills
Without trademark rules, generic manufacturers could copy the exact look of brand-name drugs. That would make it harder for patients to tell them apart. And it would let the original company keep control over the market-even after their patent expired.
Instead, the system encourages competition. Dozens of companies can make the same drug. That drives prices down. And it keeps the market honest.
Patients win. Costs drop. Safety stays high.
What’s next?
The FDA is quietly working to make generic pills look more like their brand-name versions-without breaking trademark rules. Smaller differences in color. More consistent shapes. Better labeling.
Why? Because patient confusion is still a problem. A 2023 study found that nearly 1 in 5 medication errors in older adults were linked to pill appearance changes. That’s why the FDA now recommends manufacturers aim for "similar size and shape" when possible.
But the core rule stays: no two drugs on the market can look identical. That’s the law. And it’s here to stay.
Why do generic drugs cost less if they’re the same?
Generic drugs cost less because they don’t have to repeat expensive clinical trials. The original brand-name drug already proved it’s safe and effective. Generic makers only need to show their version works the same way. That saves millions in research costs. They also don’t spend money on advertising. All that adds up to 80-85% lower prices.
Can a generic drug be less effective than the brand name?
No. The FDA requires generic drugs to have the same active ingredient, strength, dosage form, and absorption rate as the brand name. Studies show they work just as well. In fact, 89% of patients report no difference in how well their generic medicine works. If a generic fails to meet standards, the FDA pulls it off the market.
Is it safe to switch between different generic brands?
Yes. Switching between generic manufacturers is common and safe. Each one must meet the same FDA standards. The only thing that changes is the color, shape, or size. Your body still gets the same amount of medicine. If you notice any new side effects after switching, tell your doctor-but it’s likely unrelated to the switch itself.
Why does my pill look different every time I refill?
Because your pharmacy may get the generic from a different manufacturer each time. There are often 5-10 companies making the same generic drug. Each one uses its own design. That’s why your pill might be blue one month, white the next. It’s normal. Always check the label to confirm it’s the same medicine.
Do other countries have the same rules?
Most do. The U.S. is strict about trademark-based appearance rules, but countries like Canada, the UK, Australia, and New Zealand also require generics to look different from brand-name drugs. Some allow more flexibility in shape or color, but the goal is the same: prevent confusion while allowing competition.




14 Comments